 The two rows at the bottom of the periodic table are designated as the lanthanides and actinides, respectively. Vanadium, for example, shows valences of +2, +3, +4, or +5. The complicated electronic structure of the transition metals is a consequence of the similar energy of various subshells, like the 4 s and 3 d subshells, which leads to multiple valence states for single elements. This example warns you that there are exceptions to the general pattern of electronic configurations of the elements. The anomalous electronic configuration of chromium and copper is interpreted as the displacement of 1 electron from an s orbital into a d orbital these two elements have only one electron in the 4 s subshell because the second electron was promoted into a 3 d subshell. Notice the general increase in the number of electrons occupying the 3 d subshell. Figure 4 shows the valence subshell of the first series of transition metals. Each of these three rows reflects the filling of a d‐type subshell that holds up to 10 electrons. The three long rows of metallic elements in the middle of the periodic table, constituting the rectangle from scandium (21) to mercury (80), are the transition metals. The same type of subshell is used to describe the electron configurations of elements in the underlying rows. 
The six elements from boron through neon show the insertion of electrons into the lowest energy p‐type subshell. The loss of these s‐subshell valence electrons explains the common +1 and +2 charges on ions of these elements, except for helium, which is chemically inert. The two columns on the left-the alkali metals and alkaline earths-show the addition of 1 and 2 electrons into s‐type subshells. The pattern of elements in the periodic table reflects the progressive filling of electronic orbitals. Quiz: Introduction to Oxidation-Reduction Reactions.Introduction to Oxidation-Reduction Reactions. 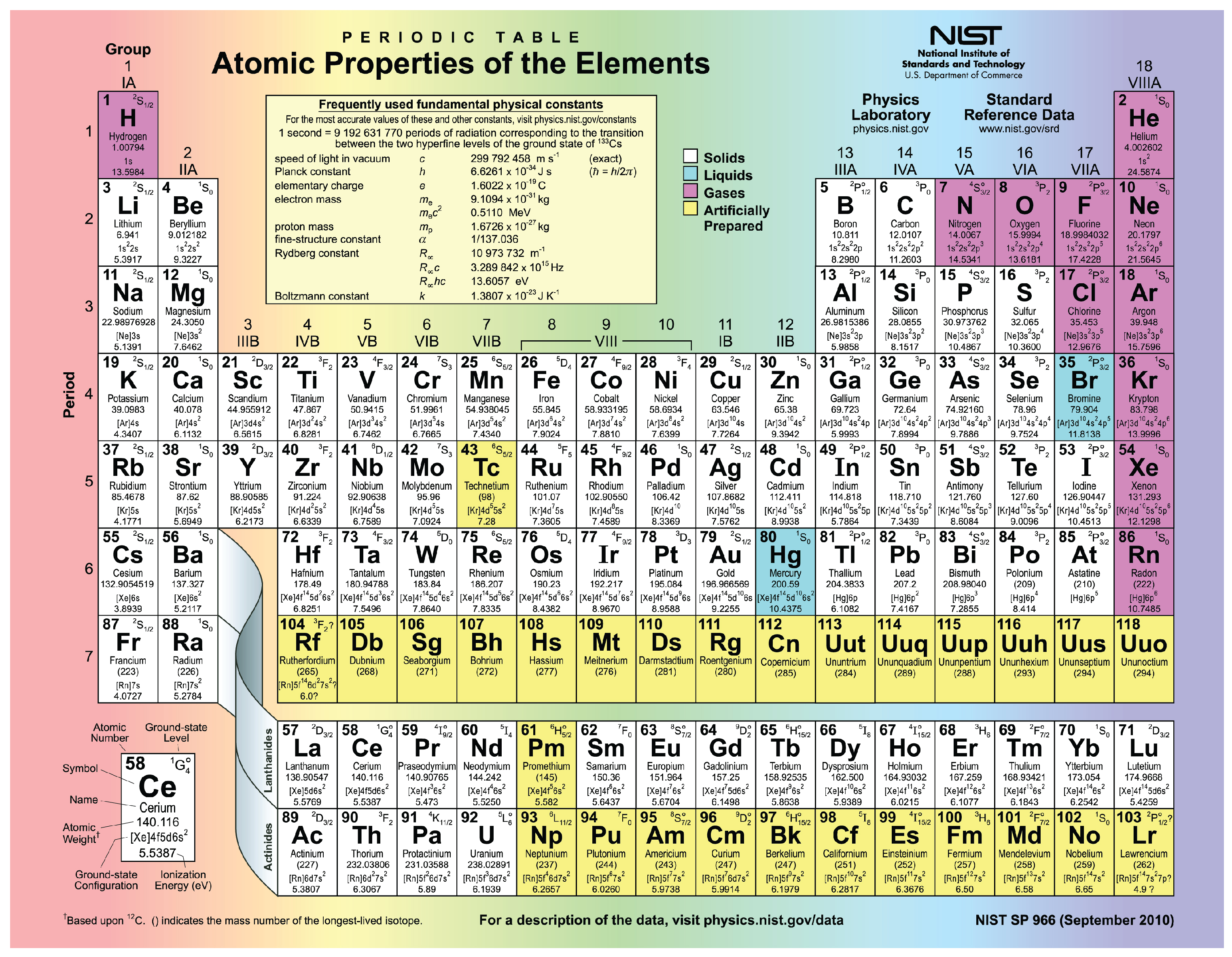
Quiz: Heat Capacities and Transformations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
